Life Sciences Is Drowning in Process Complexity
Life sciences organizations face an operational reality few other industries can match: a single New Drug Application (NDA) can exceed 100,000 pages, drug development averages 12 years and costs over $1 billion, and clinical trial teams manage hundreds of document types across safety, operational, clinical, and documentation pillars simultaneously.
This isn't just administrative burden—it's a patient safety and commercial survival issue. Medical record abstraction yields 657 errors per 10,000 fields, a 6.57% error rate that can force sample size increases of 20% or more just to preserve statistical power. The FDA issued 159 Warning Letters in 2023, and major pharmaceutical companies absorbed $18 billion in aggregate penalties over five years. Manufacturing halts alone typically erase 25% of company earnings over a decade.
Traditional RPA addressed simple copy-paste tasks, but modern life sciences demands more. Clinical narratives arrive as unstructured text. Adverse events come from patient forums, EHRs, and regulatory databases in dozens of formats. Processes span legacy LIMS, ERP, and CTMS systems that rarely communicate.
Compliance requirements—21 CFR Part 11, GxP validation, audit trail integrity—leave zero room for the rigid, exception-blind automation that basic RPA provides.
What follows covers what intelligent RPA actually automates in life sciences and what measurable outcomes organizations can expect. The convergence of regulatory pressure and widening workforce gaps has made AI-layered automation a structural necessity—not a nice-to-have.
TLDR:
- Intelligent RPA combines AI, NLP, and OCR to automate complex, judgment-intensive life sciences workflows that traditional RPA cannot handle
- High-impact use cases span clinical trials, regulatory submissions, pharmacovigilance, and supply chain—each delivering 40-90% efficiency gains
- The FDA receives over 2 million adverse event reports annually while regulatory talent gaps hit 35:1 ratios—automation is now structural, not optional
- Each day of clinical development delay costs ~$800,000 in lost sales; intelligent RPA compresses timelines at key bottlenecks
- Validated deployment requires GxP compliance and audit trails aligned with ISPE GAMP 5 and FDA CSA guidance
What Makes RPA "Intelligent" in the Life Sciences Context
Traditional RPA executes scripted, rule-based tasks: copying data between systems, filling forms, clicking through workflows. It's fast, accurate, and cheap—but only when inputs are structured, processes are predictable, and exceptions don't exist.
Intelligent RPA layers AI capabilities on top of that foundation. It integrates natural language processing (NLP) to read clinical narratives, machine learning (ML) to improve over time, optical character recognition (OCR) to extract data from PDFs and scanned documents, and computer vision to interpret images and complex layouts.
Automation Anywhere defines intelligent automation as "RPA + AI technologies" built to handle processes that rely on unstructured data, complex information sources, or workflows that don't follow fixed rules.
Why Life Sciences Needs the Intelligence Layer
Life sciences workflows expose every limitation of basic RPA:
- Regulatory complexity — submissions reference MedDRA terminology, ICH M4 module structures, and GxP requirements that demand semantic understanding, not keyword matching
- Unstructured data — adverse event narratives, patient forum posts, physician notes, and clinical trial site reports rarely arrive in clean, templated formats
- Legacy system fragmentation — LIMS, ERP, CTMS, document management platforms, and regulatory databases weren't built to integrate, so UI-based automation must adapt to each system's layout
- Zero tolerance for errors — a missed 15-day adverse event reporting deadline or a transcription error in a regulatory submission can trigger FDA Warning Letters, halt approvals, or leave safety risks undetected
Intelligent RPA handles these scenarios through AI-augmented decision-making. A traditional RPA bot can copy adverse event reports from an intake form to a database, but only if the form fields match exactly.
An intelligent RPA system reads a free-text patient forum post, extracts the medical event using NLP, maps it to MedDRA terminology, checks it against existing case reports, and routes it for pharmacovigilance review—no predefined templates required.
Maturity Stages: From Basic Bots to Intelligent Platforms
Automation in life sciences typically evolves through three stages:
- Attended/unattended RPA — Bots handle high-volume, structured tasks like populating CRFs from standardized lab results or generating batch production reports
- AI-augmented RPA — NLP and OCR enable extraction from variable formats, such as reading physician notes or processing invoices from different suppliers
- Intelligent automation platforms — End-to-end orchestration combines RPA, ML models, workflow engines, and validation frameworks to manage complex processes like regulatory submission preparation or supply chain deviation management

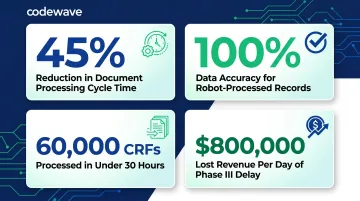
The ICON plc case study demonstrates this progression: ICON established an RPA Centre of Excellence in 2019, processed 60,000 CRFs in under 30 hours (versus over one week manually), and freed 3 FTEs per document type—achieving 45% cycle time reduction and 100% data accuracy.
Intelligent RPA does not replace scientists, pharmacovigilance professionals, or regulatory affairs specialists. It eliminates volume-intensive tasks so experts can focus on high-judgment work: interpreting safety signals, designing trial protocols, and making the regulatory strategy calls that require human expertise.
Key Processes Intelligent RPA Automates in Life Sciences
Intelligent RPA drives measurable impact across five functional domains in life sciences. The right automation priority depends on where your organization's manual burden creates the greatest compliance risk, timeline delay, or cost.
Clinical Trials and Drug Development
Clinical trial management generates massive data volumes across hundreds of sites, thousands of patients, and dozens of systems. Intelligent RPA accelerates trial timelines by:
- Automating patient data extraction from EHRs and case report forms (CRFs), reducing the 0.29%-6.57% error rates associated with manual data entry
- Populating clinical trial management systems (CTMS) with enrollment status, site performance metrics, and protocol deviation tracking
- Checking enrollment eligibility criteria by cross-referencing patient records against inclusion/exclusion rules automatically
- Monitoring adverse events in real time across trial sites, flagging serious events for immediate review
ICON plc, managing over 500 document types per trial, reduced document processing cycle time by 45% and achieved 100% data accuracy for robot-processed records. In an industry where each day of Phase III trial delay costs $55,716 in direct expenses and approximately $800,000 in lost sales, even modest timeline compression delivers measurable ROI.
Bots systematically cross-validate data from multiple trial sites, flagging discrepancies faster than any manual review cycle. This reduces data lock timelines—the critical gate before regulatory submission—and ensures data integrity issues are caught early when correction costs are lowest.
Regulatory Compliance and Submissions
Regulatory submissions represent one of the most document-intensive, error-prone processes in life sciences. Intelligent RPA automates:
- Document management for regulatory dossiers — organizing SOPs, generating audit trails, and maintaining version control across thousands of documents
- eCTD formatting and submission preparation — structuring submissions to ICH M4 module standards, creating hyperlinks and bookmarks, extracting metadata from documents
- Compliance checking — validating submissions against FDA or EMA guidelines before filing, flagging missing sections or formatting errors
- Multi-agency format adaptation — converting submissions to meet different regulatory body requirements without manual reformatting
These tasks are specific and costly: manually editing PDF properties, creating bookmark structures for 100,000-page submissions, maintaining hyperlink integrity across document versions, and ensuring metadata consistency. They consume weeks of regulatory affairs professional time and introduce errors that can trigger FDA Complete Response Letters, each delaying approval by months and costing approximately $800,000 per day in lost revenue.
Pharmacovigilance and Drug Safety Monitoring
The FDA's FAERS database contains over 28 million adverse event reports as of December 2023, with over 2 million new reports added annually. Individual Case Safety Reports drive more than 50% of postmarketing safety-related labeling changes in the US, yet pharmacovigilance teams face 35:1 job-posting-to-candidate ratios.
Intelligent RPA addresses this volume-versus-workforce mismatch by:
- Monitoring multiple data sources — EHRs, patient forums, social media, regulatory databases, and literature for adverse event signals
- Extracting key information using NLP from unstructured narratives, including event descriptions, patient demographics, drug exposure, and outcomes
- Populating standardized adverse event forms automatically, reducing what used to take days to hours
- Flagging cases for pharmacovigilance review based on seriousness criteria, regulatory reporting timelines, and signal detection algorithms
CSL Behring doubled adverse event auto-coding rates from 30% to over 60% using NLP, while Agios Pharmaceuticals achieved a 10-fold decrease in time to process Serious Adverse Event Report Forms. AstraZeneca automated reporting for 100,000 adverse events worldwide, saving millions of dollars annually.
Faster adverse event detection directly narrows the window during which a safety signal goes undetected. The Novo Nordisk FDA Warning Letter (March 2026) cited multiple violations including failure to report deaths and suicidal ideation events within mandatory 15-day deadlines—demonstrating that pharmacovigilance failures carry severe regulatory consequences.
Supply Chain and Manufacturing Operations
Pharmaceutical supply chains and manufacturing operations face unique complexity: serialization requirements, temperature-controlled distribution, batch genealogy tracking, and zero-tolerance quality standards. Intelligent RPA optimizes these operations through:
- Automated inventory reconciliation across ERP systems, warehouses, and distribution centers
- Real-time tracking of serialized products through the supply chain
- Automated reordering based on demand forecasts and safety stock levels
- Supplier compliance checks against DSCSA, EU FMD, and internal quality standards
- Bill of materials generation and batch record validation
For manufacturing, bots monitor batch records in real time, flag deviations from predefined production parameters, and generate compliance-ready manufacturing reports automatically. Over 80% of pharmaceutical manufacturing deviations are attributed to human error, and a 1% increase in deviation rate can translate into hundreds of thousands of dollars in lost yield annually.
Electronic Batch Records (EBR) integrated with intelligent RPA can reduce a 150-page paper batch record to a 3-page exception report, cutting batch review cycle time from days to hours while maintaining complete audit trail compliance.

Back-Office and Administrative Operations
While less technically complex than scientific workflows, back-office processes often consume disproportionate staff time and create compliance risk when done manually. High-value automation targets include:
- HR onboarding for regulated roles — validating credentials, conducting background checks, generating training records, and provisioning system access
- Claims and invoice processing — extracting data from invoices, matching to purchase orders, validating coding, and routing for approval
- Contract management — tracking contract terms, renewal dates, and compliance obligations
- Financial reconciliation — matching transactions across systems, identifying discrepancies, and generating audit-ready reports
- IT account provisioning — creating user accounts, assigning permissions based on role, and maintaining access logs
- Regulatory reporting dashboards — aggregating data from multiple systems to generate compliance reports
These functions benefit as much from automation as core scientific operations. Invoice processing automation achieves 50% faster cycle times, while HR onboarding automation reduces manual effort by 35% and ensures complete documentation for regulatory audits.
Why Intelligent RPA in Life Sciences Matters Right Now
Life sciences organizations are under pressure from three directions simultaneously: tightening regulations, exploding data volumes, and a workforce that cannot scale fast enough. Intelligent RPA addresses all three — not as a future investment, but as an operational necessity right now.
Regulatory Pressure Intensification
Regulatory scrutiny has intensified across every major market, with data integrity requirements under 21 CFR Part 11 and GxP compliance expanding alongside inspection frequency. The cost of falling short is measurable:
- FDA issued 159 Warning Letters in 2023
- Industry-wide regulatory fines reached $190 million in 2022
- Major pharmaceutical companies absorbed $18 billion in aggregate penalties over five years
Manual documentation isn't just inefficient—it's risky. The Novo Nordisk Warning Letter demonstrates how pharmacovigilance failures trigger regulatory action. Manufacturing halts due to GMP violations typically erase 25% of company earnings over 10 years, and GMP violations carry an average fine of $5.3 million per citation.

Intelligent RPA addresses these risks by eliminating manual transcription errors, maintaining complete audit trails, and ensuring process consistency across thousands of transactions. When every data field must be traceable and every workflow must be validated, automation provides the control and documentation that manual processes cannot.
Data Volume Explosion
Next-generation therapies are generating data complexity at a scale manual teams cannot absorb:
- Cell and gene therapy manufacturing creates unique batch genealogy tracking requirements and real-time quality parameter monitoring across highly variable processes
- Precision medicine generates multi-omic datasets that must be integrated with clinical outcomes and regulatory documentation
- Digital biomarkers from wearables produce continuous patient monitoring data that must be processed, analyzed, and incorporated into safety databases
The Cell and Gene Therapy Manufacturing Services Market is valued at $9.29 billion in 2026, growing at 16.18% CAGR, indicating the scale of this data challenge. Human teams cannot process this volume at the required speed or accuracy without automation support.
Competitive and Commercial Urgency
Time-to-market directly impacts revenue and patient access. The Tufts Center for the Study of Drug Development calculates that each day of delay in drug development equals approximately $800,000 in unrealized prescription drug sales and $55,716 in direct Phase III trial costs. For cardiovascular drugs, median daily sales reach $1.4 million; for oncology drugs, $840,000.
Intelligent RPA compresses timelines at every stage:
- Clinical development moves 45% faster through automated document processing (ICON case study)
- Regulatory approval cycles shrink as automated submission preparation turns months-long formatting tasks into days
- Supply chain response improves through real-time inventory reconciliation and automated reordering that cut stockouts
These timeline improvements translate directly into earlier revenue recognition and competitive positioning, particularly in markets with patent expiration pressures.
Workforce Sustainability
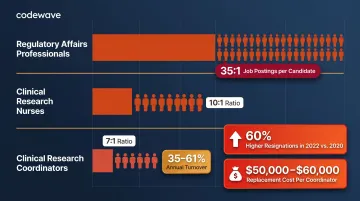
Life sciences organizations face critical talent shortages:
- Regulatory affairs professionals: 35:1 job-posting-to-candidate ratio
- Clinical research nurses: 10:1 ratio
- Clinical research coordinators: 7:1 ratio, with turnover rates of 35%-61%
Resignations among clinical research professionals with 5-10 years of tenure were 60% higher in 2022 than in 2020. Replacing a single coordinator costs $50,000-$60,000, and 95% of cancer centers reported staffing shortfalls — driving a 20% drop in trial accrual rates since January 2020.
Intelligent RPA allows the same team to handle higher volumes without proportional headcount growth, reducing burnout while maintaining compliance. It doesn't replace skilled professionals—it eliminates the volume-intensive tasks that prevent them from doing the work only they can perform.
The Business Impact: What Organizations Actually Achieve
Measurable ROI from intelligent RPA in life sciences clusters around five categories: reduced operational costs, fewer compliance penalties, faster clinical trial completion, improved pharmacovigilance response times, and supply chain cost savings.
Documented Time and Cost Savings
The ICON/UiPath case study provides the most detailed publicly available life sciences RPA outcomes:
- 45% reduction in document processing cycle time
- 2x processing speed versus human operators
- 60,000 CRFs processed in under 30 hours versus over one week manually
- 3 FTEs freed per document type from ETMF automation
- 100% data accuracy for robot-processed records (zero transcription errors)
- Single-digit thousands of hours saved across the organization
CSL Behring doubled adverse event auto-coding rates from 30% to over 60% using NLP for MedDRA mapping. Agios Pharmaceuticals achieved a 10-fold decrease in time to process Serious Adverse Event Report Forms. AstraZeneca automated reporting for 100,000 adverse events worldwide, saving millions of dollars annually.
For manufacturing, Electronic Batch Records can reduce a 150-page paper batch record to a 3-page exception report and cut batch review cycle time from days to hours. That compression translates directly into faster release timelines and improved supply chain responsiveness.
Quality Improvement and Compliance Outcomes
Beyond speed, automation eliminates manual data entry errors that create downstream compliance and quality problems. Manual medical record abstraction yields 657 errors per 10,000 fields (6.57%), while single data entry produces 29 errors per 10,000 fields (0.29%). High error rates can necessitate sample size increases of 20% or more to preserve statistical power, which drives up trial costs and extends timelines.
Automation reduces error rates to near-zero (ICON reported 100% accuracy for robot-processed records), improving:
- Regulatory submission success rates — fewer Complete Response Letters due to data quality issues
- Audit outcomes — complete, consistent audit trails across all automated processes
- GxP compliance — validated systems that execute processes identically every time, eliminating human variability
ROI Trajectory and Scaling Effects
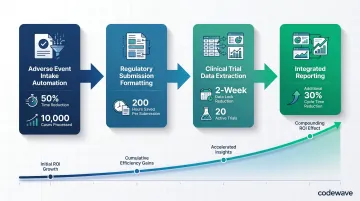
ROI improves with scale. Organizations that start with one or two automated processes and expand systematically see compounding gains as more workflows are connected. A typical scaling sequence looks like this:
- Adverse event intake automation cuts processing time by 50% across 10,000 annual cases
- Regulatory submission formatting saves 200 hours per submission across 5 annual submissions
- Clinical trial data extraction reduces data lock timelines by 2 weeks across 20 active trials
- Data from automated adverse event intake flows directly into automated regulatory reports, eliminating manual reconciliation and cutting reporting cycle time by an additional 30%
Early deployments do more than deliver immediate savings — they build the validation frameworks and integration patterns that cut implementation time for every subsequent automation. Organizations that start now compress years of capability-building into months.
Building Your Intelligent RPA Roadmap in Life Sciences
Getting intelligent RPA right in life sciences starts before you write a single line of automation logic. Process selection, validation planning, and sequencing decisions made upfront determine whether deployment succeeds or stalls in rework.
Process Discovery and Prioritization
Start by identifying processes that meet these criteria:
- High volume — thousands of transactions per year, creating measurable time burden
- Rule-driven but complex — processes that follow logic but require interpretation of variable inputs
- Error-prone — current manual execution produces quality issues or compliance gaps
- Compliance-critical — failures create regulatory risk, patient safety issues, or audit findings
Conduct formal process mining or assessment before selecting automation targets. Map current workflows end-to-end, identify bottlenecks, quantify error rates, and calculate cost of delay. Prioritize processes where automation delivers the highest ROI with the lowest implementation complexity.
High-priority candidates typically include:
- Adverse event intake and coding
- Regulatory submission document assembly
- Clinical trial site data reconciliation
- Batch record review and deviation management
- Invoice and purchase order processing
Lower-priority candidates include:
- Processes requiring frequent human judgment calls
- Workflows with highly variable inputs that resist standardization
- Systems with unstable interfaces that change frequently
- Low-volume processes where manual effort is minimal
Validation and Compliance Considerations
Life sciences automation faces unique validation requirements under GxP. Every automated system must undergo:
- Computer system validation (CSV) following ISPE GAMP 5 Second Edition risk-based principles
- Audit trail integrity complying with 21 CFR Part 11 requirements for electronic records and signatures
- Change management that maintains validated state through updates and patches
- Integration testing with legacy systems (LIMS, ERP, CTMS, document management) to ensure data integrity across platforms
- Data security controls for patient data, clinical trial information, and proprietary research
GAMP 5 Second Edition (July 2022) and FDA Computer Software Assurance guidance (February 2026) both promote risk-based, critical-thinking approaches to validation—shifting away from exhaustive documentation toward exception-based reporting for low-risk functions. This evolution supports scalable automation deployment, but implementation must still follow validated workflows.
Implementation Approach and Timeline
With compliance requirements defined, the implementation approach determines how quickly teams reach validated, scalable outcomes. Deep life sciences domain knowledge and technical automation expertise must work together — generic RPA implementation without regulatory context leads to non-compliant systems and costly rework.
Critical success factors include:
- Implementation teams must understand GxP requirements, regulatory submission workflows, pharmacovigilance processes, and clinical trial operations
- CSV requirements, audit trail design, and risk assessment must be integrated into initial architecture — not retrofitted after development
- Start with a single, well-defined process, validate thoroughly, achieve measurable outcomes, then expand systematically
- Train end users, document new workflows, update SOPs, and maintain validated state through ongoing operations
Organizations that cannot afford lengthy, speculative implementation cycles need approaches that move from validated process design to working automation in days rather than months. Codewave's QuantumAgile™ methodology does exactly that: it simulates multiple implementation paths, identifies the highest-value deployment sequence, and ships validated automation incrementally while maintaining GxP compliance throughout.
Frequently Asked Questions
What is the difference between traditional RPA and intelligent RPA in life sciences?
Traditional RPA handles structured, rule-based tasks using scripted bots that execute predefined workflows. Intelligent RPA adds AI/ML, NLP, and OCR to handle unstructured data, process exceptions, and judgment-intensive workflows—making it suitable for complex life sciences environments where data arrives in variable formats and processes require interpretation.
Which life sciences processes are best suited for intelligent RPA?
Pharmacovigilance, regulatory submissions, clinical trial data management, supply chain reconciliation, and back-office operations deliver the highest ROI. Prioritize processes where data volumes are high, manual errors carry compliance risk, and timeline compression impacts patient access or revenue recognition.
How does intelligent RPA handle unstructured data like adverse event narratives?
NLP components read free-text patient narratives, extract relevant medical events, map them to standardized terminology (such as MedDRA), and route cases for human review, without requiring structured input formats. This enables processing of social media posts, patient forum discussions, and physician notes.
What are the regulatory compliance requirements when implementing RPA in life sciences?
Automated systems in GxP environments must undergo computer system validation (CSV), maintain complete audit trails, and comply with 21 CFR Part 11. Validated, life-sciences-specific implementation is critical; generic RPA deployments without regulatory context fail audits and require costly rework.
Can intelligent RPA integrate with existing systems like LIMS, ERP, and CTMS?
Yes. Intelligent RPA connects disparate systems without requiring replacement, acting as a bridge that reads from and writes to existing platforms through UI interaction or APIs. This preserves existing system investments while eliminating manual data transfer.
How long does it take to see ROI from intelligent RPA in a life sciences organization?
Initial deployments in well-defined processes (such as adverse event intake or document formatting) show measurable time savings within weeks. Enterprise-wide ROI compounds as more workflows are connected over 6-18 months, with scaling effects accelerating later-stage deployments.


